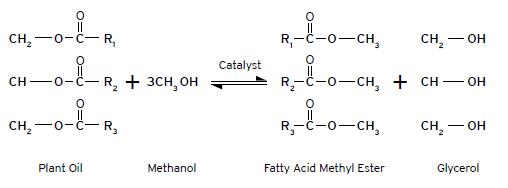
During the process of transesterification, an alcohol (such as methanol) reacts with the triglyceride oils contained in plant oils, animal fats or recycled greases to form fatty acid alkyl esters (biodiesel) and glycerin. The reaction requires heat and a strong base catalyst such as sodium hydroxide or potassium hydroxide. The simplified chemical reaction is shown below.
Some types of feedstocks require pretreatment before they can go through the transesterification process. Feedstocks with less than 4% free fatty acids such as most plant oils and some food-grade animal fats do not require pretreatment. However, feedstocks with more than 4% fatty acids require pretreatment using an acid esterification process. These include inedible animal fats and recycled greases. In this pretreatment step, the feedstock is reacted with an alcohol (like methanol) in the presence of a strong acid catalyst (like sulfuric acid) to convert the free fatty acids into biodiesel. The remaining triglycerides are then converted to biodiesel through the usual transesterification reaction.
Triglycerides + Free Fatty Acids + Alcohol = Alkyl Esters + Triglycerides
The complete process for the production of methyl ester from plant oil and other feedstocks therefore involves basically five steps: acid esterification, transesterification, methanol recovery, biodiesel refining, and glycerin refining.
Acid Esterification. The oil feedstock containing more than 4% free fatty acids is usually pretreated using an acid esterification process to increase the yield of biodiesel. The feedstock is first filtered and then pre-processed to remove water and other contaminants such as unwanted solids. The pretreated oil is then fed to the acid esterification process. The catalyst, sulfuric acid, is dissolved in methanol and then mixed with the pretreated oil. The mixture is heated and stirred, and the free fatty acids are converted to biodiesel. Once the reaction is complete, it is dewatered and then fed to the transesterification process.
Transesterification. The plant oil, which contains less than 4% free fatty acids, is first filtered and then pre-processed to remove water and other contaminants. The pretreated oil is then fed directly to the transesterification process along with any products of the acid esterification process. The catalyst, potassium hydroxide, is dissolved in methanol and then mixed with the pretreated oil. If an acid esterification process is used, then additional alkaline catalyst must be added to neutralize any excess acid remaining from that step. Once the reaction is complete, the major co-products, biodiesel and glycerin, are separated into two layers.
Methanol recovery. The methanol is usually removed immediately after the biodiesel and glycerine have been separated. This is done to prevent the reaction from reversing itself. The recovered methanol is cleaned and recycled back to the beginning of the process.
Biodiesel refining. Once separated from the glycerin, the biodiesel goes through a series of cleaning-up or purification steps to remove excess alcohol, residual catalyst and soaps. These consist of multistage washings with clean water. The product biodiesel is then dried and sent to storage. If required, the product biodiesel can be further refined through an additional distillation step to produce a colorless, odorless, zero-sulfur, and premium quality biodiesel.
Glycerin refining. The crude glycerin from the transesterification process may be recovered or used in a fuel blend for steam production. The crude glycerin contains unreacted catalyst and soaps that must be neutralized with an acid. The water and alcohol are also removed to produce 50%-80% crude glycerin. The remaining contaminants include unreacted fats and oils. In large biodiesel plants, the glycerin can be further purified through a series of unit operations to produce a product of 99% or higher purity. This purified product is suitable for use in the pharmaceutical and cosmetic industries.